Summary
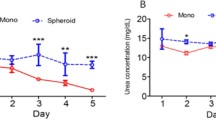
Primary hepatocytes cultured as monolayers or as spheroids were studied to compare the effects of four different culture media (Williams' E, Chee's, Sigma Hepatocyte, and HepatoZYME medium). Rat hepatocytes were cultured as conventional monolayers for 3 d or as spheroids for 2 wk. For spheroid formation a method was employed that combined the use of a nonadherent substratum with rotation of cultures. Hepatocyte integrity and morphology were assessed by light and electron microscopy and by reduced glutathione content. Hepatocyte function was measured by albumin secretion and 7-ethoxycoumarin metabolism. Chee's medium was found to be optimal for maintenance of hepatocyte viability and function in monolayers, but it failed to support spheroid formation. For spheroid formation and for the maintenance of spheroid morphology and function, Sigma HM was found to be optimal. These results demonstrate that the medium requirements of hepatocytes differ markedly depending on the culture model employed. Spheroid culture allowed better preservation of morphology and function of hepatocytes compared with conventional monolayer culture. Hepatocytes in spheroids formed bile canaliculi, and expressed an actin distribution resembling that found in hepatocytes in vivo. Albumin secretion was maintained at the same level as that found during the first d in primary culture, and 7-ethoxycoumarin metabolism was maintained over 2 wk in culture at approximately 30% of the levels found in freshly isolated hepatocytes. The improved morphology and function of hepatocyte cultures as spheroids may provide a more appropriate in vitro model for certain applications where the maintenance of liver-specific functions in long-term culture is crucial.
Similar content being viewed by others
References
Arterburn, L. M.; Zurlo, J.; Yager, J. D.; Overton, R. M.; Heifetz, A. H. A morphological study of differentiated hepatocytes in vitro. Hepatology 22:175–187; 1995.
Asano, K.; Koide, N.; Tsuji, T. Ultrastructure of multicellular spheroids formed in primary culture of adult rat hepatocytes. J. Clin. Electron Microsc. 22:243–252; 1989.
Balavoine, S.; Feldemann, G.; Lardeux, B. Regulation of RNA degradation in cultured rat hepatocytes: effects of specific amino acids and insulin. J. Cell. Physiol. 156:56–62; 1993.
Ballard, F. J.; Wong, S. S.; Knowles, S. E.; Partrige, N. C.; Martin, T. J.; Wood, C. M.; Gunn, J. M. Insulin inhibition of protein degradation in cell monolayers. J. Cell. Physiol. 105:335–346 1980.
Battle, T.; Maguire, T.; Moulsdale, H.; Doyle, A. Progressive maturation resistance to microcystin-LR cytotoxicity in two different hepatospheroidal models. Cell Biol. Toxicol. 15:3–12; 1999.
Bradford, M. M. A rapid and sensitive method for quantification of microgram quantities of protein utilising the principle of protein-dye binding. Anal. Biochem. 72:248–254; 1976.
Coecke, S.; Rogiers, V.; Bayliss, M., et al. The use of long-term hepatocyte cultures in detecting induction of drug metabolising enzymes: the current status. ATLA 27:579–638; 1999.
Cravedi, J. P.; Paris, A.; Monod, G.; Devaux, A.; Flouriot, G.; Valotaire, Y. Maintenance of cytochrome P450 content and phase I and phase II enzyme activities in trout hepatocytes cultured as spheroidal aggregates. Comp. Biochem. Physiol. C: Pharmacol. Toxicol. Endocrinol. 113:241–246; 1996.
Dahn, M. S.; Hsu, C. J.; Lange, M. P.; Kimball, S. R.; Jefferson, L. S. Factors affecting secretory protein production in primary culture of rat hepatocytes. Proc. Soc. Exp. Biol. Med. 203:38–44; 1993.
Dilworth, C.; Hamilton, G. A.; George, E.; Timbrell, J. A. The use of liver spheroids as an in vitro model for studying induction of the stress response as a marker of chemical toxicity. Toxicol. In Vitro 14:169–176; 2000.
Donato, M. T.; Gómez-Lechón, M. J.; Castell, J. V. Rat hepatocytes cultured on a monkey kidney cell line: expression of drug-biotransformation activities by hepatocytes. Toxicol. In Vitro 5:435–483; 1991.
Dunn, J. C. Y.; Tompkins, R. G.; Yamush, M. L. Long-term in vitro function of adult hepatocytes in collagen sandwich configuration. Biotechnol. Prog. 7:237–245; 1991.
Flaim, K. E.; Hutson, S. M.; Lloyd, C. E.; Taylor, J. M.; Shiman, R.; Jefferson, L. S. Direct effect of insulin on albumin gene expression in primary cultures of adult rat hepatocytes. Am. J. Physiol. 249:E447-E453; 1985.
Freshney, R. I. Culture of animal cells: a manual of basic technique. 3rd ed. New York: Wiley; 1994:71–103.
Gerlach, J. C.; Klooel, K.; Muller, C.; Schnoy, N.; Smith, M. D.; Neuhaus, P. Hepatocyte aggregate culture technique for bioreactors in hybrid liver support systems. Int. J. Artif. Organs 16:843–846; 1993.
Guguen-Guillouzo, C.; Clement, B.; Baffet, G.; Beumont, C.; Morel-Chany, E.; Glaise, D.; Guillouzo, A. Maintenance and reversibility of active albumin secretion by adult rat hepatocytes cocultured with another liver epithelial cell type. Exp. Cell Res. 143:47–54; 1983.
Guguen-Guillouzo, C.; Gripon, P.; Vandenberghe, Y.; Lamballe, F.; Ratanasavanh, D.; Guillouzo, A. Hepatotoxicity and molecular aspects of hepatocyte function in primary culture. Xenobiotica 18:773–783; 1988.
Ham, R. G.; McKeehan, W. L. Media and growth requirements. In: Jakoby, W. B.; Pastan, I. H., ed. Methods in enzymology. Vol. 58. New York: Academic Press; 1979:44–94.
Hissin, P. J.; Hilf, R. A fluorimetric method for determination of oxidized and reduced glutathione in tissues. Anal. Biochem. 89:187–206; 1975.
Hofmann, W.; Guder, W. G. A diagnostic programme for quantitative analysis of proteinuria. J. Clin. Chem. Clin. Biochem. 27:589–600; 1989.
Hutson, S. M.; Stinton-Fischer, C.; Shiman, R.; Jefferson, L. S. Regulation of albumin synthesis by hormones and amino acids in primary culture of rat hepatocytes. Am. J. Physiol. 252:E291-E298; 1987.
Jauregui, H. O.; McMillan, P. N.; Driscoll J.; Naik, S. Attachment and long term survival of adult rat hepatocytes in primary monolayer cultures: comparison of different substrata and tissue culture medium formulations. In Vitro Cell. Dev. Biol. 22:13–22; 1986.
Koebe, H. G.; Pahernik, S.; Eyer, P.; Schildberg, F. W. Collagen gel immobilization: a useful cell culture technique for long-term metabolism studies on human hepatocytes. Xenobiotica 24:95–107; 1994.
Koide, N.; Sakaguchi, K.; Koide, Y., et al. Formation of multicellular spheroids composed of adult rat hepatocytes in dishes with positively charged surfaces and under other nonadherent environments. Exp. Cell Res. 186:227–235; 1990.
Kojima, T.; Mochizuki, C.; Tobioka, H.; Saitoh, M.; Takahashi, S.; Mitika, T.; Mochizuki, Y. Formation of actin filament networks in cultured rat hepatocytes treated with DMSO and glucagon. Cell Struct. Funct. 22(2):269–278; 1997.
Laishes, B. A.; Williams, G. M. Conditions affecting primary cell cultures of functional adult rat hepatocytes. II. Dexamethasone enhanced longevity and maintenance of morphology. In Vitro 12:821–832; 1976.
Landry, J.; Bernier, D.; Ouellet, C.; Goyette, R.; Marceau, N. Spheroidal aggregate culture of rat liver cells: histotypic reorganization, biomatrix deposition, and maintenance of functional activities. J. Cell Biol. 101:914–923; 1985.
Lang, F. S.; Zdunek, D.; Engel, D.; Ziegenhorn, J. Immunoturbidimetric determination of urinary albumin on Hitachi analysers [abstract]. Clin. Chem. 36:998; 1990.
LeCluyse, E. L.; Bullock, P. L.; Parkinson, A. Strategies for restoration and maintenance of normal hepatic structure and function in long-term cultures of rat hepatocytes. Adv. Drug Deliv. Rev. 22:133–186; 1996.
Lee, J.; Morgan, J. R.; Tomkins, R. G.; Yarmush, M. L. The importance of proline on long-term hepatocyte function in collagen gel sandwich configuration: regulation of protein secretion. Biotechnol. Bioeng. 40:298–305; 1992.
Li, A. P.; Colburn, S. M.; Beck, D. J. A simplified method for the culturing of primary adult rat and human hepatocytes as multicellular spheroids. In Vitro Cell. Dev. Biol. 28A:673–677; 1992.
Musat, A. I.; Sattler, C. A.; Sattler, G. L.; Pitot, H. C. Reestablishment of cell polarity of rat heaptocytes in primary cultures. Hepatology 18(1):198–205; 1993.
Peshwa, M. V.; Wu, F. J.; Sharp, H. L.; Cerra, F. B.; Hu, W. S. Mechanistics of formation and ultrastructural evaluation of hepatocyte spheroids. In Vitro Cell. Dev. Biol. 32A:197–203; 1996.
Phillips, M. J. Biology and pathology of actin in the liver. In: Arias, I. A.; Boyer, J. L.; Fausto, N.; Jackoby, W. B.; Schachter, D.; Schafritz, D. A., ed. The liver biology and pathology, 3rd ed. New York: Raven Press; 1994:19–32.
Roberts, R. A.; Soames, A. R. Hepatocyte spheroids: prolonged hepatocyte viability for in vitro modeling of nongenotoxic carcinogenesis. Fundam. Appl. Toxicol. 21:149–158; 1993.
Rogiers, V.; Blaauboer, B.; Maurel, P.; Phillips, P.; Shephard, E. Hepatocytebased in vitro models and their application in pharmacotoxicology. Toxicol. In Vitro 9:685–694; 1995.
Runge, D.; Runge, D. M.; Jager, D., et al. Serum-free, long-term cultures of human hepatocytes: maintenance of cell morphology, transcription factors, and liver-specific functions. Biochem. Biophys. Res. Commun. 1:46–53; 2000.
Sakai, Y.; Naruse, K.; Nagashima, I.; Muto, T.; Suzuki, M. Large-scale preparation and function of porcine hepatocyte spheroids. Int. J. Artif. Organs 19:294–301; 1996.
Sato, Y.; Ochiya, T.; Yasuda, Y.; Matsubara, K. A new three-dimensional culture system for hepatocytes using reticulated polyurethane. Hepatology 19:1023–1028; 1994.
Schwarze, P. E.; Solheim, A. E.; Seglen, P. O. Amino acid and energy requirements for rat hepatocytes in primary culture. In Vitro 18:43–54; 1982.
Seglen, P. O. Preparation of rat liver cells. Exp. Cell Res. 76:25–30; 1973.
Seglen, P. O.; Gordon, P. B.; Poli, A. Amino acid inhibition of autophagic/lysosomal pathway of protein degradation in isolated hepatocytes. Biochim. Biophys. Acta. 630:103–118; 1980.
Sidhu, J. S.; Omiecinski, C. J. Modulation of xenobiotic-inducible cytochrome P450 gene expression by dexamethasone in primary rat hepatocytes. Pharmacogenetics 5:24–36; 1995.
Skett, P.; Roberts, P. Effect of culture medium on the maintenance of steroid metabolism in cultured adult rat hepatocytes. In Vitro Toxicol. 7:261–267; 1994.
Tong, J. Z.; DeLagausie, P.; Furlan, V.; Cresteil, T.; Bernard, O.; Alvarez, F. Long-term culture of adult rat hepatocyte spheroids. Exp. Cell Res. 200:326–332; 1992.
Ueno, K.; Endoh, K.; Miyashita, A.; Takezawa, Y.; Mori, Y.; Satoh, T. An alternative to hepatotoxicity testing using multicellular spheroids composed of rat liver cells. In Vitro Toxicol. 7:351–355; 1994.
Walker, T.; Rhodes, P. C.; Westmoreland, C. The differential cytotoxicity of methotrexate in rat hepatocyte monolayer and spheroid cultures. Toxicol. In Vitro 14:475–485; 2000.
Walker, T.; Woodrooffe, A.; Westmoreland, C. Testosterone metabolism in rat hepatocyte spheroid cultures: time-course of metabolic activity and cytochrome P450 induction. ATLA 27(Suppl.): 287; 1999.
Walsh, J. S.; Patanalla, J. E.; Halm, K. A.; Facchine, K. L. An improved HPLC assay for the assessment of liver slice metabolic viability using 7-ethoxycoumarin. Drug Metab. Dispos. 23:869–874; 1995.
Watts, P.; Smith, M. D.; Edwards, I.; Zammit, V.; Brown, V.; Grant, H. The influence of medium composition on the maintenance of cytochrome P-450, glutathione content and urea synthesis: a comparison of rat and sheep primary hepatocyte cultures. J. Hepatol. 23:605–612; 1995.
Wu, F. J.; Friend, J. R.; Hsioa, C. C.; Zilliox, M. J.; Ko, W. J.; Cerra, F. B.; Hu, W. S. Effecient assembly of rat hepatocyte spheroids for tissue engineering applications. Biotech. Bioeng. 50:404–415; 1996.
Yasuura, S.; Ueno, T.; Watanabe, S.; Hirose, M.; Namihisa, T. Immunocytochemical localisation of myosin in normal and phalloidin-treated rat hepatocytes. Gastroenterology 97:982–989; 1989.
Yumoto, A. U.; Watanabe, S.; Hirose, M.; Kitamura, T.; Yamaguchi, Y.; Sato, N. Structural and functional features of bile canaliculi in adult rat hepatocyte spheroids. Liver 16:61–66; 1996.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hamilton, G.A., Westmoreland, C. & George, E. Effects of medium composition on the morphology and function of rat hepatocytes cultured as spheroids and monolayers. In Vitro Cell.Dev.Biol.-Animal 37, 656–667 (2001). https://doi.org/10.1290/1071-2690(2001)037<0656:EOMCOT>2.0.CO;2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1290/1071-2690(2001)037<0656:EOMCOT>2.0.CO;2